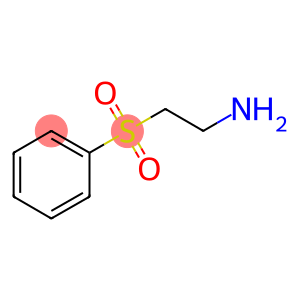
2-AMINOETHYLPHENYLSULPHONE HYDROCHLORIDE
Ethanamine,2-(phenylsulfonyl)-, hydrochloride (1:1)
CAS: 38752-48-6
Molecular Formula: C8H12ClNO2S
2-AMINOETHYLPHENYLSULPHONE HYDROCHLORIDE - Names and Identifiers
2-AMINOETHYLPHENYLSULPHONE HYDROCHLORIDE - Physico-chemical Properties
| Molecular Formula | C8H12ClNO2S |
| Molar Mass | 221.7 |
| Melting Point | 155-155.5 °C |
| Storage Condition | Room Temprature |
2-AMINOETHYLPHENYLSULPHONE HYDROCHLORIDE - Risk and Safety
| Risk Codes | 41 - Risk of serious damage to eyes |
| Safety Description | S26 - In case of contact with eyes, rinse immediately with plenty of water and seek medical advice. S39 - Wear eye / face protection. |
2-AMINOETHYLPHENYLSULPHONE HYDROCHLORIDE - Introduction
Ethanamine, 2-(phenylsulfonyl)-, hydrochloride (1:1)(Ethanamine, 2-(phenylsulfonyl)-, hydrochloride (1:1)) is an organic compound with the chemical formula C8H11NO2S · HCl. The following is a description of its nature, use, preparation and safety information:
Nature:
-Appearance: White crystalline powder or crystal.
-Melting point: about 90-94 ℃.
-Solubility: Soluble in water and alcohol solvents.
Use:
- Ethanamine,2-(phenylsulfonyl)-, hydrochloride (1:1) is widely used as an intermediate in chemical research.
-It is often used as an important reagent and catalyst in organic synthesis.
Preparation Method:
- Ethanamine,2-(phenylsulfonyl)-, hydrochloride (1:1) can be prepared by the reaction of phenylthionyl chloride and ethylamine.
Safety Information:
- Ethanamine,2-(phenylsulfonyl)-, hydrochloride (1:1) has not yet produced significant toxicity and harm to humans and the environment.
-However, safety measures should be taken during handling and use, such as wearing appropriate personal protective equipment (such as gloves, glasses, etc.), operating in a well-ventilated area, and avoiding direct contact with skin and eyes.
-If taken by mistake or in contact with eyes, rinse immediately with plenty of water and seek medical advice.
Please note that the use and handling of chemical substances should follow the appropriate safe operating guidelines, and ensure that they are used with detailed information and professional guidance.
Nature:
-Appearance: White crystalline powder or crystal.
-Melting point: about 90-94 ℃.
-Solubility: Soluble in water and alcohol solvents.
Use:
- Ethanamine,2-(phenylsulfonyl)-, hydrochloride (1:1) is widely used as an intermediate in chemical research.
-It is often used as an important reagent and catalyst in organic synthesis.
Preparation Method:
- Ethanamine,2-(phenylsulfonyl)-, hydrochloride (1:1) can be prepared by the reaction of phenylthionyl chloride and ethylamine.
Safety Information:
- Ethanamine,2-(phenylsulfonyl)-, hydrochloride (1:1) has not yet produced significant toxicity and harm to humans and the environment.
-However, safety measures should be taken during handling and use, such as wearing appropriate personal protective equipment (such as gloves, glasses, etc.), operating in a well-ventilated area, and avoiding direct contact with skin and eyes.
-If taken by mistake or in contact with eyes, rinse immediately with plenty of water and seek medical advice.
Please note that the use and handling of chemical substances should follow the appropriate safe operating guidelines, and ensure that they are used with detailed information and professional guidance.
Last Update:2024-04-10 22:29:15
Supplier List
Spot supply
Product Name: 2-(Phenylsulfonyl)ethanamine Hydrochloride Visit Supplier Webpage Request for quotationCAS: 38752-48-6
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: 2-AMINOETHYLPHENYLSULFONE HYDROCHLORIDE Request for quotation
CAS: 38752-48-6
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
CAS: 38752-48-6
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
Product Name: 2-AMINOETHYLPHENYLSULFONE HYDROCHLORIDE Visit Supplier Webpage Request for quotation
CAS: 38752-48-6
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 38752-48-6
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
Spot supply
Product Name: 2-(Phenylsulfonyl)ethanamine Hydrochloride Visit Supplier Webpage Request for quotationCAS: 38752-48-6
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: 2-AMINOETHYLPHENYLSULFONE HYDROCHLORIDE Request for quotation
CAS: 38752-48-6
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
CAS: 38752-48-6
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
Product Name: 2-AMINOETHYLPHENYLSULFONE HYDROCHLORIDE Visit Supplier Webpage Request for quotation
CAS: 38752-48-6
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 38752-48-6
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
View History


